AKEEGA® delivered a meaningful* rPFS benefit and delayed time to symptomatic progression1
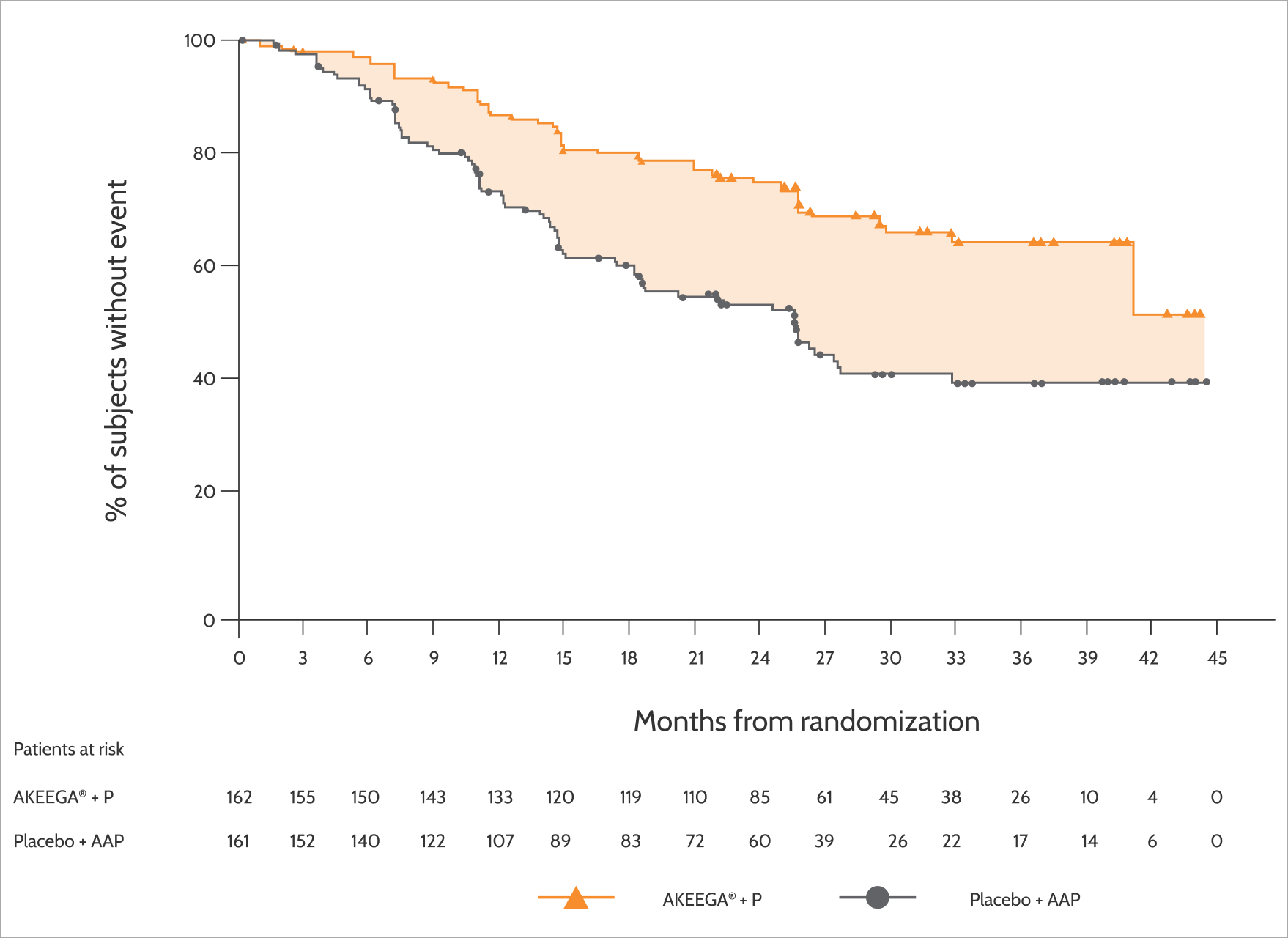
Progression-free survival
AKEEGA® delivered a meaningful* reduction in the risk of radiographic progression or death1
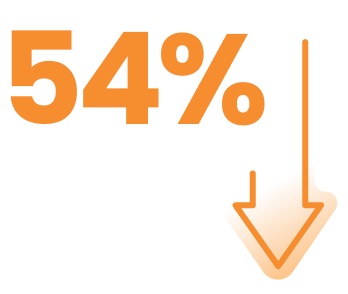
reduction in risk of radiographic progression or death vs standard of care (SoC‡)
HR, 0.46 (95% CI, 0.32-0.66)§
Primary endpoint:
Radiographic progression-free survival (rPFS)1†

reduction in risk of radiographic progression or death vs standard of care (SoC‡)
HR, 0.46 (95% CI, 0.32-0.66)§
*Defined as a favorable impact on patient feeling, function, or survival.2
†Investigator-assessed.
‡SoC = abiraterone acetate + prednisone.1
§Calculated using an unstratified Cox proportional hazards model.
In the HRRm patient population
- A statistically significant improvement in rPFS with AKEEGA® + P vs placebo + AAP was seen in the overall population of patients with HRRm1
- In an exploratory analysis of the subgroup of 373 patients with non-BRCA2 m mCSPC, the rPFS HR was 0.88 (95% CI: 0.63, 1.24), indicating that improvement in the overall population was primarily attributable to the results seen in the BRCA2 m mCSPC subgroup1
ADDITIONAL ENDPOINTS
Interim analysis (BRCA2 m subgroup): additional endpoints
AKEEGA® delayed the time to symptomatic progression vs SoC1
59%improvement
in time to symptomatic progression vs SoC (HR, 0.41 95% CI, 0.26-0.65)
AKEEGA® reduced the incidence of death vs SoC1
78%of patients
were alive at follow-up vs 66% in the SoC arm
Study design
AMPLITUDE: the first trial to evaluate a PARPi + NHT combination in BRCA2 m mCSPC1,3
Prior to study entry, patients were permitted1:

≤6 cycles of docetaxel

≤6 months of ADT

≤45 days of AAP
AMPLITUDE Study Design1,3,4
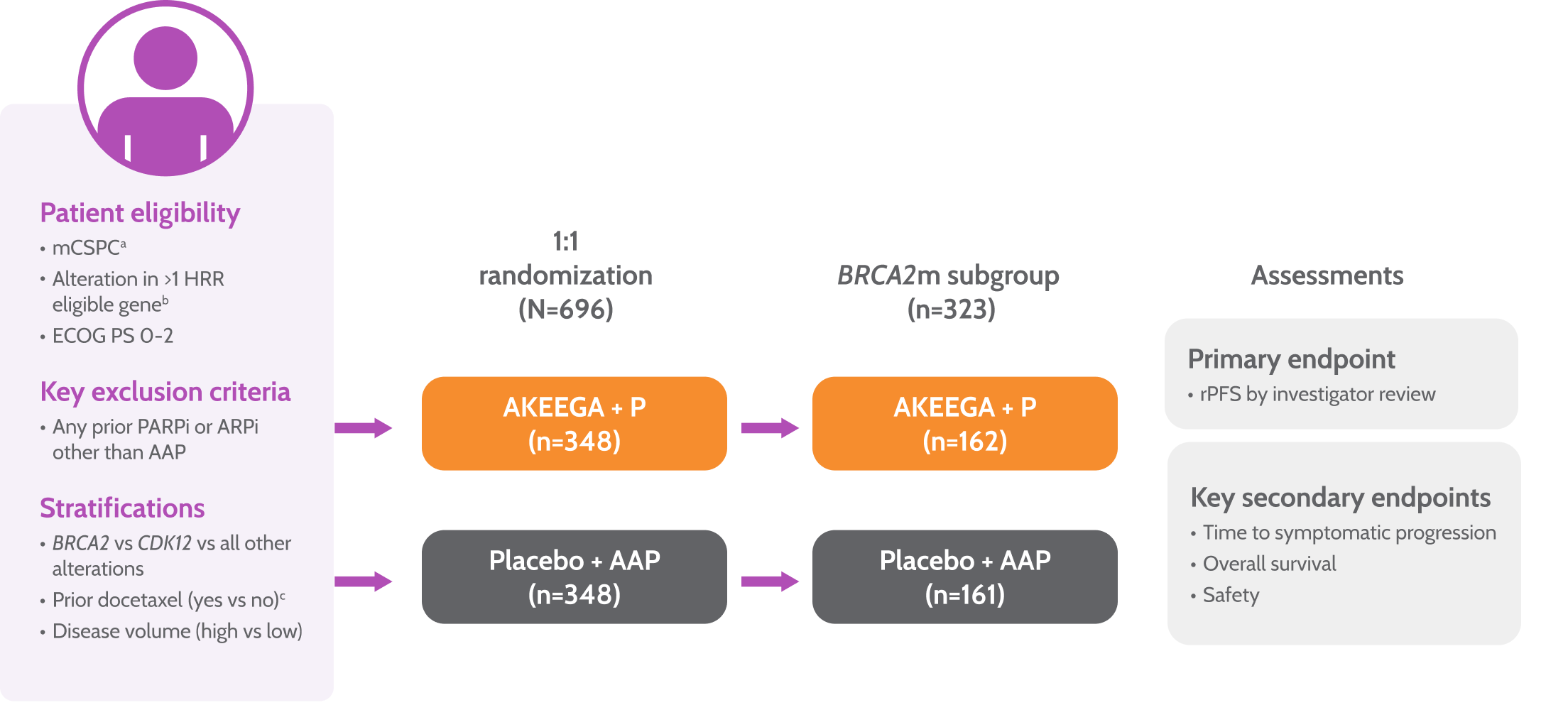
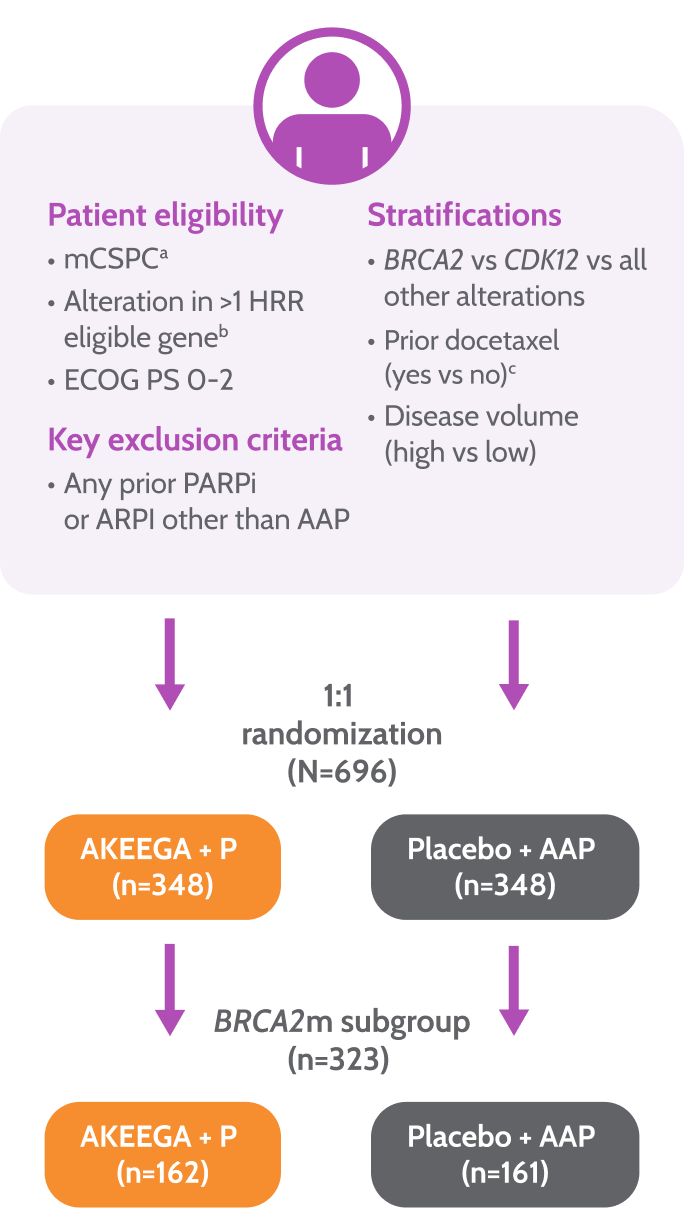
Primary endpoint
• rPFS by central review
Key secondary endpoints
• Time to symptomatic progression
• Overall survival
• Safety
aPatients with lymph node–only disease were not eligible.
bHRR gene panel was fixed prior to trial initiation based on MAGNITUDE trial and external data from the published literature.
cLast dose of docetaxel ≤3 months prior to randomization.
- First and final rPFS analysis and first interim analysis of time to symptomatic progression and overall survival for the study population. Clinical data cutoff: January 7, 2025. Median follow-up: 30.8 months3
AMPLITUDE baseline characteristics were well-balanced between
treatment arms3,4
At baseline, among patients in the AKEEGA® treatment arm4:
77%
had high disease volume
~50%
had lymph node metastases
16%
had visceral metastases
Baseline Characteristics (all patients)4
| AKEEGA® + P(n=348) | Placebo + AAP(n=348) | |||
|---|---|---|---|---|
| Age, median (range), y | 68 (40-88) | 67 (40-92) | ||
| PSA at initial diagnosis median (range), (ng/mL) | 112 (0.1-17475)a | 102 (0.1-15900)b | ||
| ECOG PS, n (%) | 0≥1 | 242 (70)106 (30) | 218 (63)130 (37) | |
| Gleason score at initial diagnosis, n (%) | ≥8 | 276 (79) | 262 (75) | |
| Metastatic stage at diagnosis, n (%) | M1 (Synchronous) | 301 (86) | 302 (87) | |
| Disease volume, n (%) | High | 269 (77) | 271 (78) | |
| Prior docetaxel use in mCSPC, n (%) | 54 (16) | 54 (16) | ||
| Site of metastasesc, n (%) | Bone onlyVisceralLymph nodes | 146 (42)57 (16)173 (50) | 154 (44)d54 (16)d161 (46)d | |
| BRCA alteration, n (%) | 191 (55) | 196 (56)d | ||
an=258.
bn=275.
cNon-mutually exclusive.
dn=347.
AAP, abiraterone acetate + prednisone; ARPi, androgen receptor pathway inhibitor; BRCA2 m, BRCA2 gene-mutated; BRCA m, BRCA gene-mutated; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; HRR, homologous recombination repair; mCSPC, metastatic castration-sensitive prostate cancer; NHT, novel hormonal therapy; OS, overall survival; P, prednisone; PARPi, poly-ADP ribose polymerase inhibitor; PS, performance status; PSA, prostate-specific antigen; rPFS, radiographic progression-free survival; SoC, standard of care.
References:
- AKEEGA® [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc.
- Walton MK, Powers JH, Hobart J, et al. Clinical outcome assessments: conceptual foundation—report of the ISPOR Clinical Outcomes Assessment—emerging good practices for outcomes research task force.Value Health. 2015;18(6):741-752.
- Attard G, Agarwal N, Graff JN, et al. Niraparib and abiraterone acetate plus prednisone for HRR-deficient metastatic castration-sensitive prostate cancer: a randomized phase 3 trial. Nat Med. 2025. doi:10.1038/s41591-025-03961-8
- Data on file. Janssen Biotech, Inc.
